Carried by: FAVAMULTI
References:
Clinician(s): Sophie DUPUIS-GIROD
Biologist(s): Maud TUSSEAU, Florence COULET
Presentation
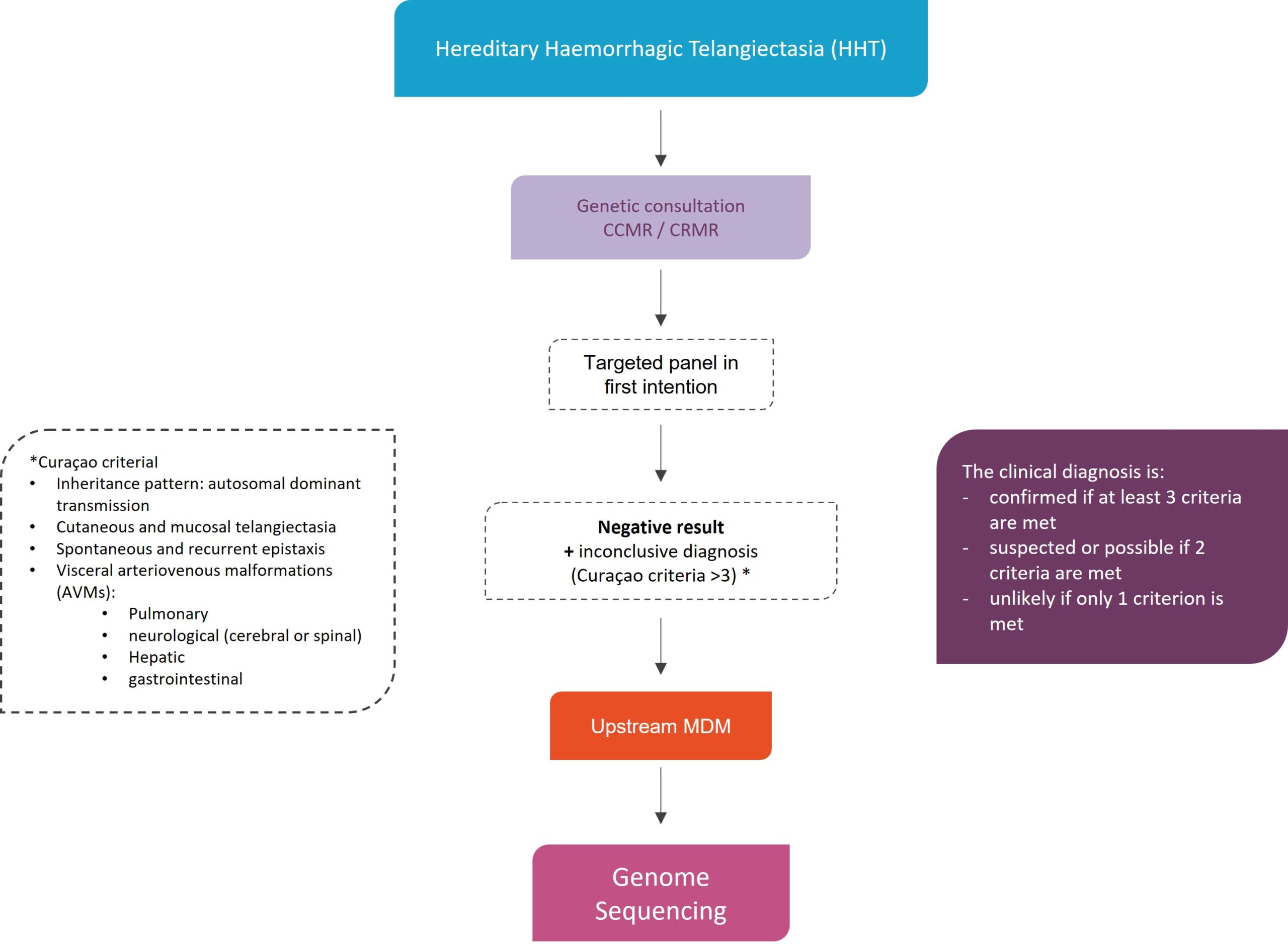
Currently, national diagnosis and treatment protocols specify that testing for pathogenic variants in the ACVRL-1, ENG and SMAD4 genes must be offered to all patients with a clinically confirmed or probable diagnosis, as well as to relatives of affected individuals in whom the familial pathogenic variant responsible for HHT has been identified, in accordance with bioethics laws.
In patients with definite clinical diagnosis, the use of currently developed gene panels allows pathogenic variant to be identified in more than 90% of cases.
Criteria before considering a discussion in MDM-FMG
- Definitive clinical diagnosis (Curaçao criteria ≥ 3)
Shovlin C et al, 2000; Am J Med Genet 91:66-67*
- No mutation identified by the current panel
* Curaçao criteria
- Hereditary nature: autosomal dominant transmission
- Cutaneous and mucosal telangiectasia
- Spontaneous and recurrent epistaxis
- Visceral arteriovenous malformations (AVMs)
- pulmonary
- neurological cerebral or medullary
- hepatic
- digestive
The clinical diagnosis is:
- certain if at least 3 criteria are present,
- suspected or possible if 2 criteria are present,
- unlikely if only 1 criterion is present.
Genome Sequencing in diagnostic strategy
MDM
Type of the MDM
City of the coordinator
Name, first name, and email of the contact
MDM-FMG Hereditary Haemorrhagic Telangiectasia (HHT)
National
Lyon