Presentation
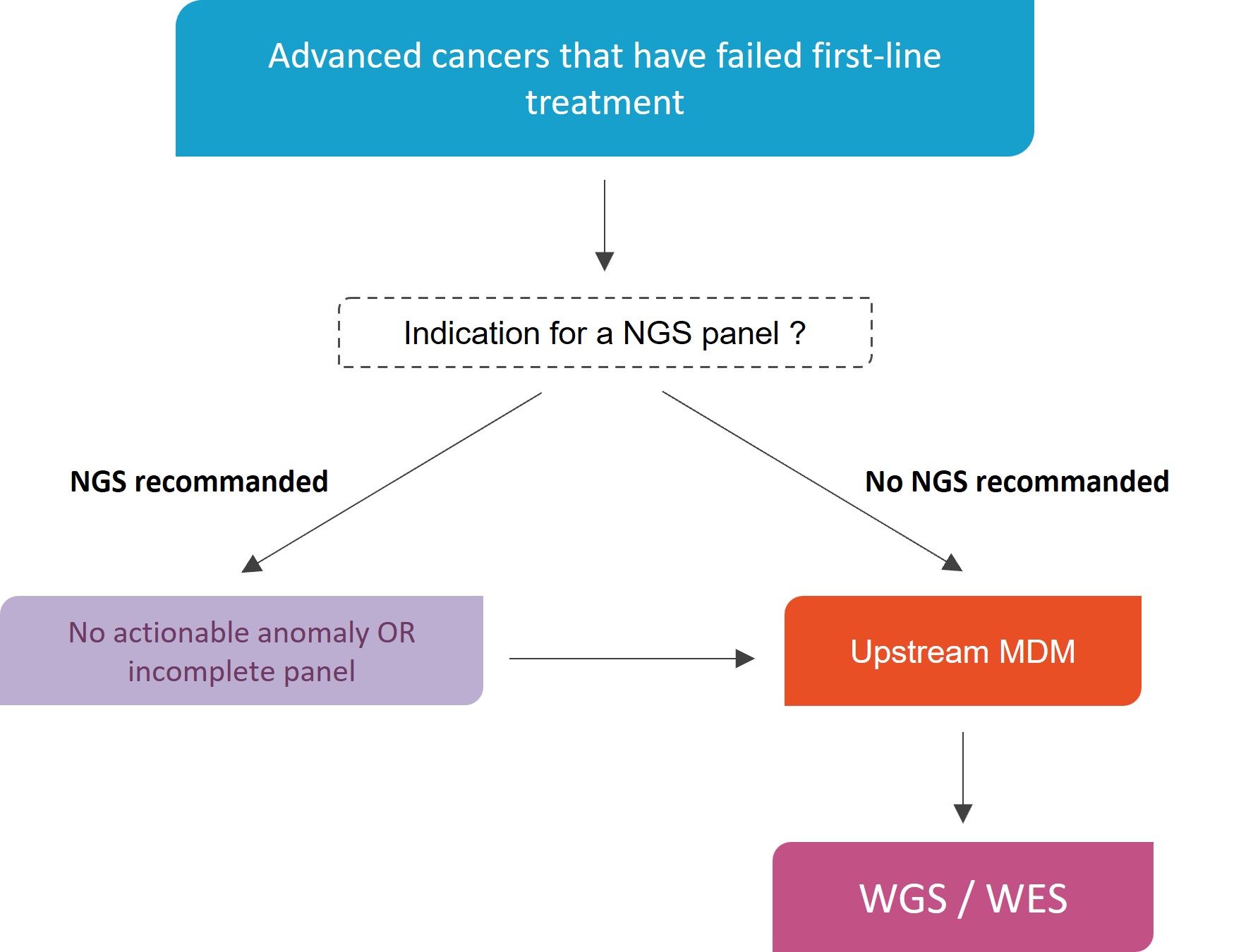
Advanced cancers that have failed first-line treatment and may benefit from targeted molecular therapy as part of a clinical trial (PRIMO)
Criteria before considering a discussion in MDM-FMG
- Advanced cancers with available material (recent biopsy or frozen material available initially)
- Life expectancy of at least 6 months (2 months between the upstream FMG2025 multidisciplinary consultation meeting and the downstream FMG2025 multidisciplinary consultation meeting)
- After therapeutic failure of first-line treatment or subsequent lines of treatment
- AND with a recommended NGS panel result in the tumour type showing no targetable alteration or no recommendation for an NGS panel
Genome Sequencing in diagnostic strategy
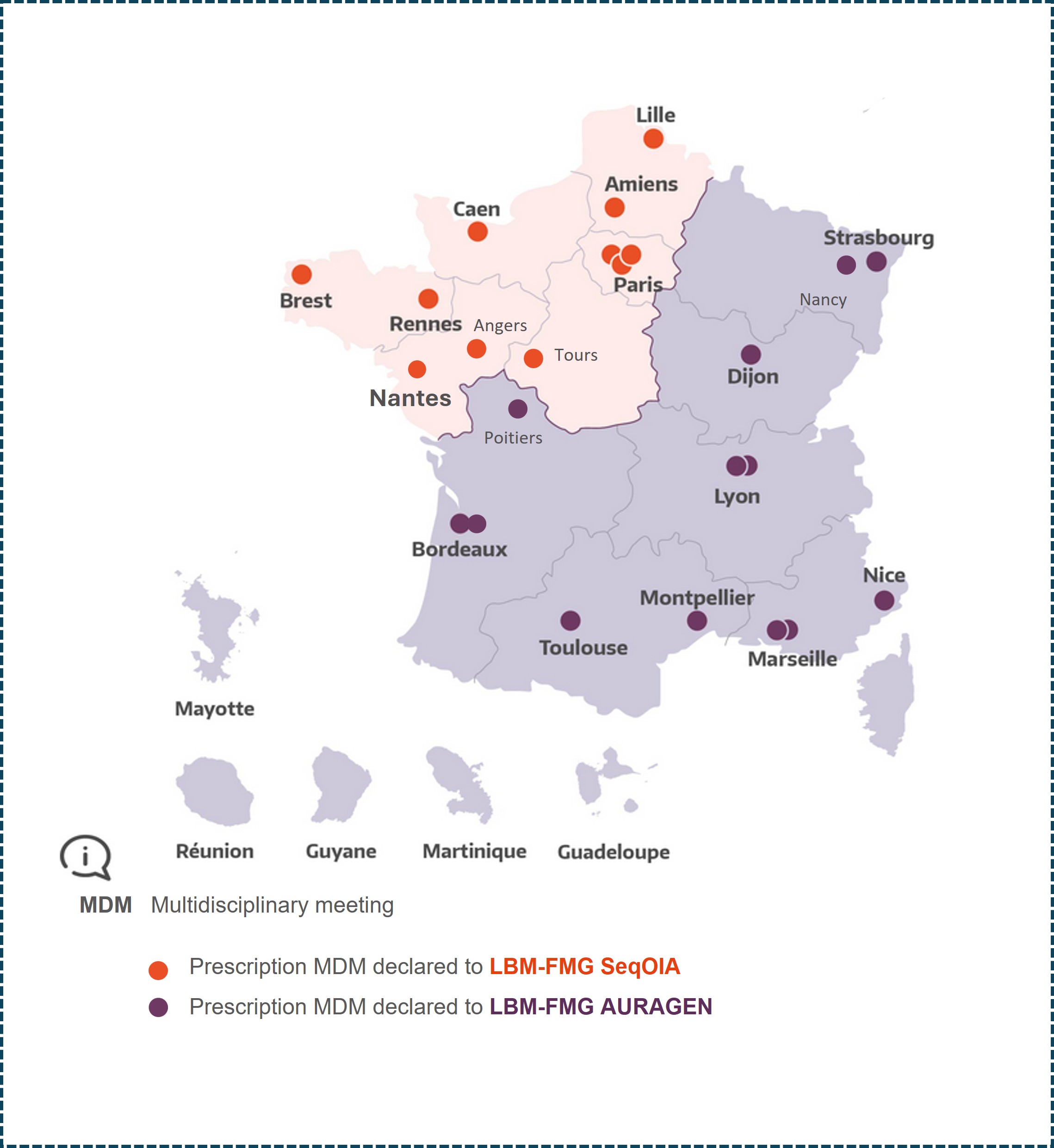
MDM FMG Solid Tumors adults Curie Paris
Sarah WATSON
RCP moléculaire équipe de coordination
MDM FMG Solid Tumors adults Gustave Roussy Villejuif
Claudio NICOTRA
MDM FMG Solid Tumors adults Rennes
Thierry Lesimple
Julien Edeline
Thibault De La Motte Rouge
Thierry Fest
Alexandra Lespagnol
Florian Estrade
MDM FMG Solid Tumors adults Brest
MDM FMG Solid Tumors adults Angers
MDM FMG Solid Tumors adults Nantes
MDM FMG Solid Tumors adults Caen
MDM FMG Solid Tumors adults Amiens
Bruno CHAUFFERT
Claire POULET
Julie DREMAUX
MDM FMG Solid Tumors adults Lille
MDM FMG Solid Tumors adults Strasbourg
Philippe BARTHELEMY
MDM FMG Solid Tumors adults Nancy
Dr Jean-Marie RAVEL
Dr Laëtitia LAMBERT
Dr Ludovic MANSUY
MDM FMG Solid Tumors adults APHM Marseille
Pascale TOMASINI
MDM FMG Solid Tumors adults IPC Marseille
François BERTUCCI
MDM FMG Solid Tumors adults Nice
Esma SAADA-BOUZID
MDM FMG Solid Tumors adults Montpellier
Stanislas Quesada
MDM FMG Solid Tumors adults Toulouse
Carlos GOMEZ-ROCA
MDM FMG Solid Tumors adults Bergonié Bordeaux
Antoine ITALIANO
MDM FMG Solid Tumors adults CHU Bordeaux
Pr MERLIO Jean-Philippe
MDM FMG Solid Tumors adults CHU Poitiers
Camille EVRARD