Carried by: SFCE ("French Society for the Fight Against Cancer and Leukaemia in Children and Adolescents")
References:
Clinician(s): B. GEOERGER, G. SCHLEIERMACHER, N. CORRADINI, N. ANDRÉ, M. STRULLU, S. DUCASSOU, A. PETIT
Biologist(s): L. LACROIX, G. PIERRON, P. NAIBO, H. CAVE, H. LAPILLONE
Presentation
Patient (child, adolescent, young adult) with paediatric cancer or leukaemia whose treatment has failed: refractory or relapsed disease, potentially eligible for a clinical trial.
Criteria before considering a discussion in MDM-FMG
- Patient potentially eligible for experimental treatment or clinical trial
- Good performance status and life expectancy > 3 months
- Availability of a frozen tumour sample in the event of relapse/progression:
- either a new sample obtained by biopsy or surgery or bone marrow aspiration
- or ‘archived’ material from a procedure performed optimally within the last 3 months
- Constitutional material (blood or bone marrow at complete remission) available
- Not applicable for certain pathologies (Hodgkin’s disease; not contributive for analysis)
MDM
Type of the MDM
City of the coordinator
Name, first name, and email of the contact
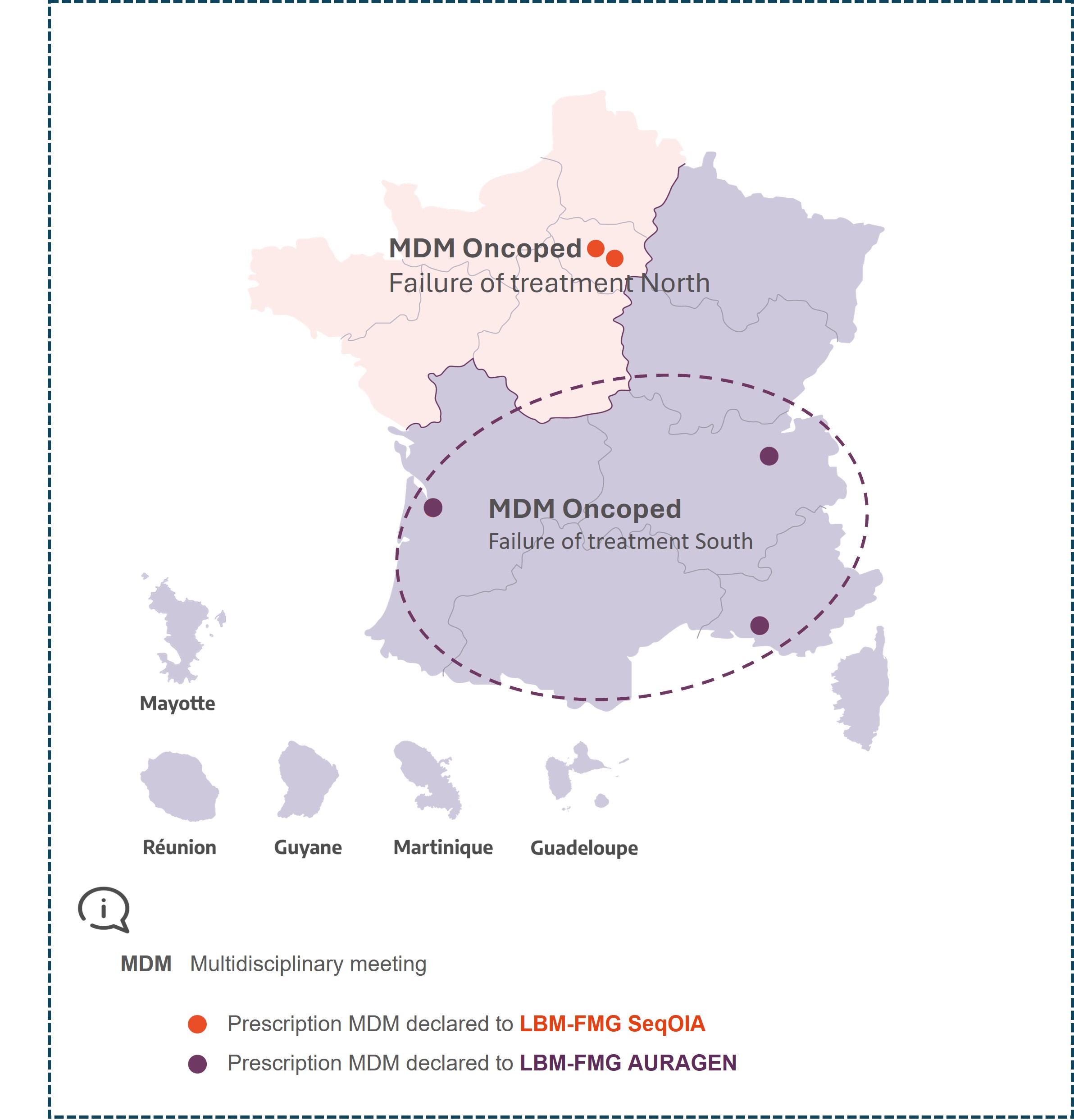
MDM Oncoped Failure of treatment North
Interregional
Villejuif/Paris
Solid tumors :
Birgit GEOERGER
Birgit.geoerger@gustaveroussy.fr
Gudrun SCHLEIERMACHER
gudrun.schleiermacher@curie.fr
Leukaemias :
Marion STRULLU
Arnaud PETIT
MDM Oncoped Failure of treatment South
Interregional
Lyon, Marseille, Bordeaux
Solid tumors :
Nadege CORRADINI
Nicolas ANDRÉ
Leukaemias :
Stéphane DUCASSOU